 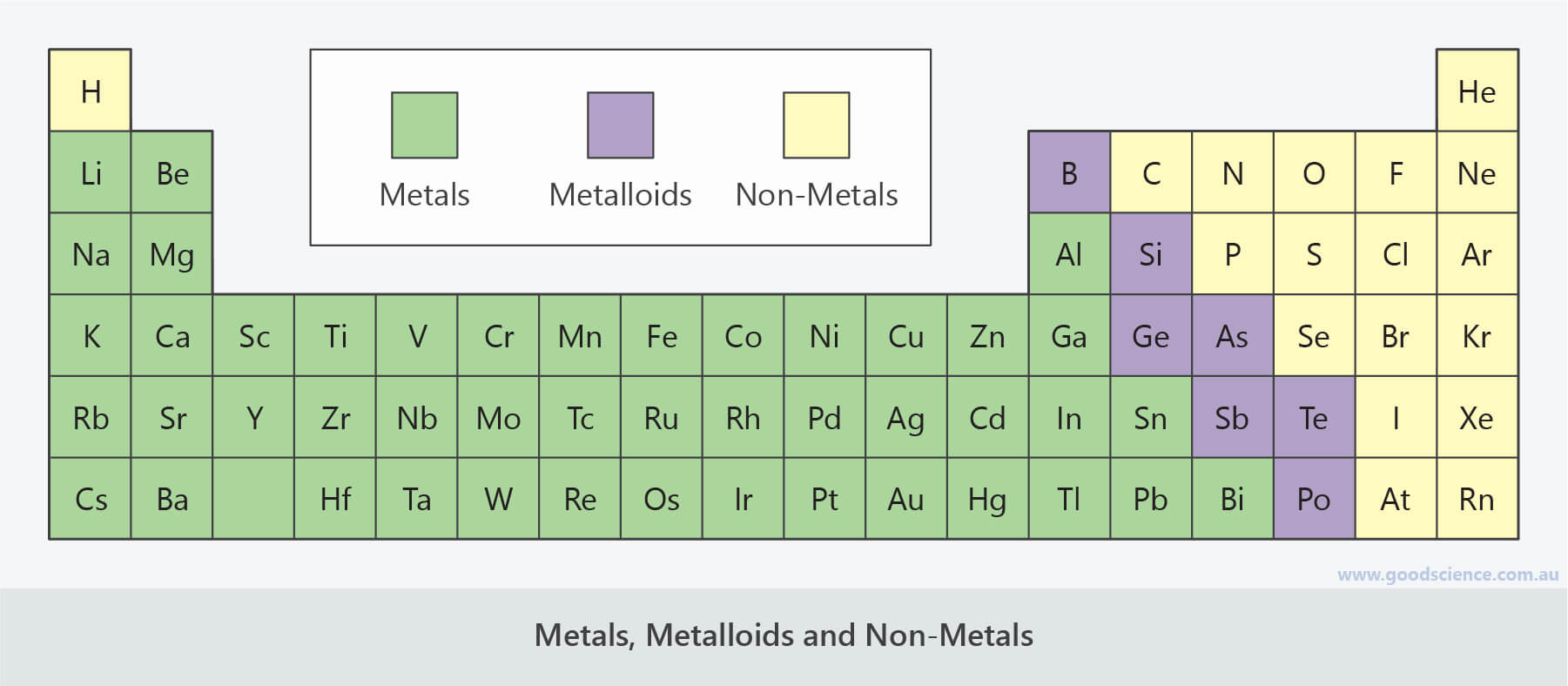 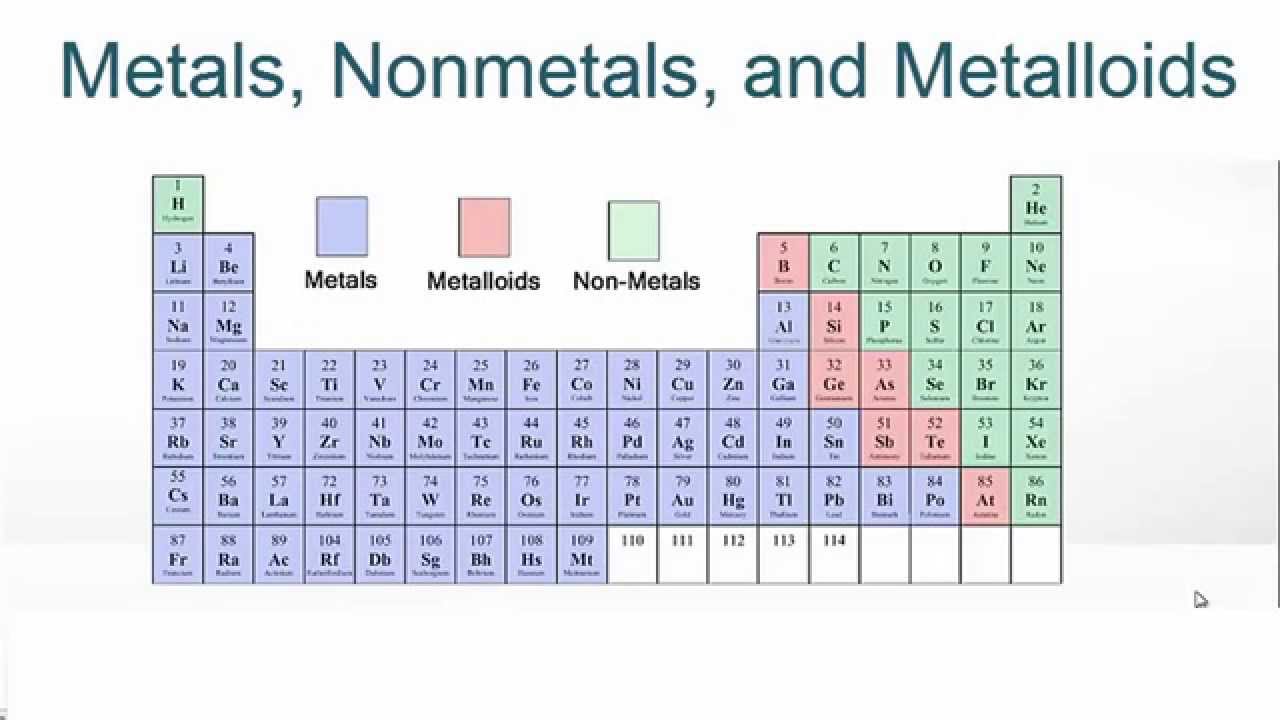
They range from colorless gases like hydrogen to shiny crystals like iodine are usually lighter (less dense) than metals and brittle or crumbly if solid. Nonmetals are chemical elements that mostly lacks distinctive metallic properties. Nonmetals in their periodic table context For nonmetallic substances, see materials science. For the use of the term nonmetal in astronomy, see nonmetal (astrophysics). Conductivity increases with temp.This article is about a class of two dozen or so chemical elements.Boron (B), Silicon (Si), Germanium (Ge), Arsenic (As), Antimony (Sb), Tellurium (Te), Polonium (Po), Astatine (At).Has appearance of metal, but behaves chemically like nonmetal.Crystal structure in which each atom surrounded by 8-12 nearest neighbors (metallic bonds between atoms).Good conductors of heat and electricity.Physical Behavior Metals Nonmetals Poor conductors Brittle and nonductile in solid state Show no metallic luster May be transparent or translucent Low density Gases, liquids, or solids Form molecules that consist of atoms covalently bonded noble gases monatomic Low ionization potentials, form cations by loss of electrons.Have 1-5 electrons in valence, usually not more than 3.React with other metals, giving metallic compounds.React with O, F, H, and other nonmetals, giving ionic compounds.Form oxides that react with water to give hydroxides.Poorer conductor of heat and electricity than metal Ĭhemical Behavior Metals Nonmetals Oxidizing agents (except noble gases) Form oxides that react with water to give acids Form acidic hydroxides React with O, F, H, and other nonmetals, giving covalent compounds React with metals, giving ionic compounds Higher electronegativities Usually have 4-8 electrons in valence High electron affinities, form anions by accepting electrons in valence (except noble gases).Found in stair shaped section of periodic table.Nonmetal Oxide + Base ––––> Salt + Water.Tend to gain electrons when react with metals.Dissolve in basic solution to form salts. 
Oxides acidic, dissolve in water to form acids (acid rain).Completely nonmetal compounds are molecular substances.Gain electrons when react with metals to form noble gas electron configuration.7 Diatomic (Gas: H2, N2, O2, F2, Cl2 Liquid: Br2 Solid: I2).Metal Oxide + Water ––––> Metal Hydroxide.Metal + Nonmetal ––––> Ionic substance.Not naturally found uncombined in nature.Bond with other elements since there is only one e- in outer shell.React with acids to form salt and water.Metal oxides basic, dissolve in water to form metal hydroxides (due to oxide ion).Compounds with nonmetals are ionic (Ex: oxides).Alkali, Alkaline Earth, Transition, and Other.Transition metals more than one positive ion.Noble Gas Electron Configuration (Alkali 1+, Alkaline Earth 2+).Nonmetallic Character- “the extent…” nonmetals.Metallic Character-the extent to which an element exhibits the physical and chemical properties of metals.Metalloids-between metals and nonmetals Ĭharacteristic Properties Metals Nonmetals No luster, various colors Solids brittle, hard and soft Poor conductors heat and electricity Oxides molecular substances form acidic solutions Anions or oxyanions in aqueous solution Cannot change shape without breaking Low melting point.No elements exist naturally as individual atoms EXCEPT noble gases.7.6 Metals, Nonmetals, and Metalloids By: Monique Briones, Marie Leibfreid, Bryanne Vollmer, Richard Wang 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
